Find out why oil does not mix in water and why water evaporates faster than oil, and what factors are responsible for these phenomena.
Oil and water are two substances that just don’t seem to get along. If you’ve tried to mix them in a salad dressing or witnessed the aftermath of an oil spill, you know that they simply refuse to mix.
But have you ever wondered why this happens and why oil does not mix with water? It turns out that there is a scientific explanation for this phenomenon.
The properties of oil and water are very different, and their molecular structures are incompatible with each other.
In this article, we will look at the scientific explanation of why oil does not mix in water and why water evaporates faster than oil, and how this knowledge can be applied to various industries, from cosmetics to environmental cleanup.
Whether you’re a science enthusiast or just curious about the world around you, this fascinating topic is sure to pique your interest and make you understand the complexity of nature in a deeper way. So let’s dive in and discover the science behind this common yet mysterious phenomenon.
Why does oil not mix with water?
Properties of water and oil
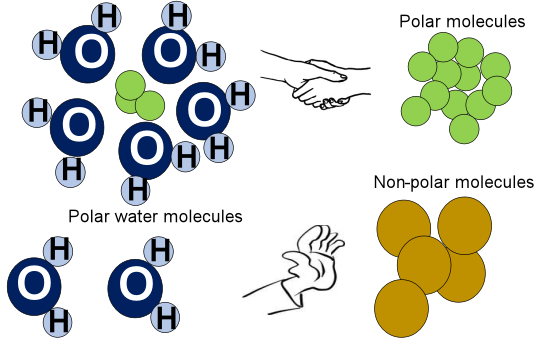
Water is a polar molecule, which means that it has both a positive and a negative end. This is due to the arrangement of the hydrogen and oxygen atoms in the molecule; the oxygen atom is more electronegative than the hydrogen atoms, causing an uneven distribution of charge throughout the molecule. This causes the water molecules to be attracted to each other by intermolecular forces known as hydrogen bonds.
Oil, on the other hand, is a non-polar substance. This means that it has no areas with positive or negative charge. Oil is made up of long chains of hydrocarbons, which are molecules made up of hydrogen and carbon atoms bonded together. Since the distribution of electrons in these molecules is uniform, there is no charge separation and no attraction between different oil molecules.
The differences between the properties of water and oil are very important in understanding why they do not mix.
Why is oil hydrophobic?
One of the key reasons why oil does not mix in water is that oil is hydrophobic, which means it repels water. This is because water molecules are more attracted to each other by hydrogen bonds than to oil molecules. When water and oil mix, the water molecules group together, leaving the oil molecules to form their own separate clusters. This is called phase separation.

Why does oil not dissolve in water?
The hydrophobic nature of oil is also the reason why oil and water do not dissolve in each other. Dissolution occurs when one substance breaks down into individual molecules or ions and is evenly distributed in another substance. Because oil molecules are larger and more complex than water molecules, they cannot dissolve in water.
The role of intermolecular forces
One of the key factors that determine whether two substances will mix is the strength of the intermolecular forces between them. Intermolecular forces are the forces of attraction or repulsion that exist between molecules. In the case of water, the hydrogen bonds between molecules are relatively strong, which means that water molecules are attracted to each other and tend to stick together.
On the other hand, oil molecules are held together by much weaker intermolecular forces, such as the London dispersion forces. These forces are much weaker than hydrogen bonds, which means that oil molecules cannot compete with water molecules for the attention of other water molecules.
Chemical structure and polarity
Another key factor that determines whether two substances will mix is their chemical structure and polarity. As we have already discussed, water is a polar molecule, which means that it has both a positive and a negative end. This polarity is due to the arrangement of hydrogen and oxygen atoms in the molecule.
Oil, on the other hand, is non-polar, which means that it has no areas with positive or negative charge. This is because oil is made up of long chains of hydrocarbons, which are non-polar molecules.
The polarity of water and the non-polarity of oil make these two substances incompatible with each other. Because of the different charges, water molecules tend to stick together and repel oil molecules.
The importance of surface tension
Surface tension is another important factor that determines whether two substances will mix. Surface tension is the force that causes the surface of a liquid to contract and form a barrier against the outside world.
Water has a relatively high surface tension, which means it tends to form droplets and resist spreading. Oil, on the other hand, has a lower surface tension, which means it tends to spread and form a thin film.
When water and oil mix, the high surface tension of the water causes it to form droplets, which are then repelled by the low surface tension of the oil. This causes the two substances to separate into separate layers.
The Building Blocks of Life: What is a Genome?
The physics of emulsification
Although oil and water do not normally mix, under certain conditions it is possible to create stable mixtures of the two substances. This process is called emulsification.
Emulsions are mixtures of two immiscible liquids, such as oil and water, that are stabilized by an emulsifier. Emulsifiers work by reducing the surface tension between the two liquids, allowing them to mix more easily.
Emulsions can be created in a variety of ways, including mechanical mixing, high-pressure homogenization, and the use of surfactants. Emulsions are used in a wide range of applications, from food and cosmetics to pharmaceuticals and industrial processes.
Why does water evaporate faster than oil?
Oil is a liquid, and it evaporates, but at a much slower rate than water. The rate of evaporation of a liquid depends on its volatility, which is determined by its vapor pressure. The vapor pressure is the pressure exerted by the molecules of a liquid when they are released into the air.
Water has a much higher vapor pressure than oil, so it evaporates much faster. In addition, oil is less polar than water and therefore does not form hydrogen bonds as easily, which also contributes to slower evaporation.

Applications of oil-water separation
Oil-water separation has important applications in a variety of industries, from cosmetics production to environmental cleanup. In the cosmetics industry, for example, oil-water separation is necessary to create stable emulsions that are used in products such as lotions and shampoos.
In the environmental industry, oil-water separation is essential for cleaning up oil spills. When an oil spill occurs, the oil and water separate naturally, with the oil rising to the surface. This allows the oil to be localized and removed from the water.
Future research and development
Although we understand the scientific basis of why oil and water do not mix, there is still much to learn about this amazing phenomenon. Researchers are constantly exploring new ways to create stable emulsions and improve oil spill response efficiency.
Advances in nanotechnology and materials science are also opening up new opportunities to create new emulsifiers and develop new separation methods.
Conclusion.
Oil and water may seem like simple substances, but their properties and behavior are actually quite complex. The inability of these two liquids to mix can be frustrating at times, but that’s what makes them so interesting.
By understanding why oil and water don’t mix, we can develop new technologies and processes that will improve our lives and protect the environment. Whether you’re a scientist, engineer, or just a person interested in the natural world, learning about oil-water separation is sure to spark your interest and imagination.
